 
The siemens, S, is the derived unit of electrical conductance. 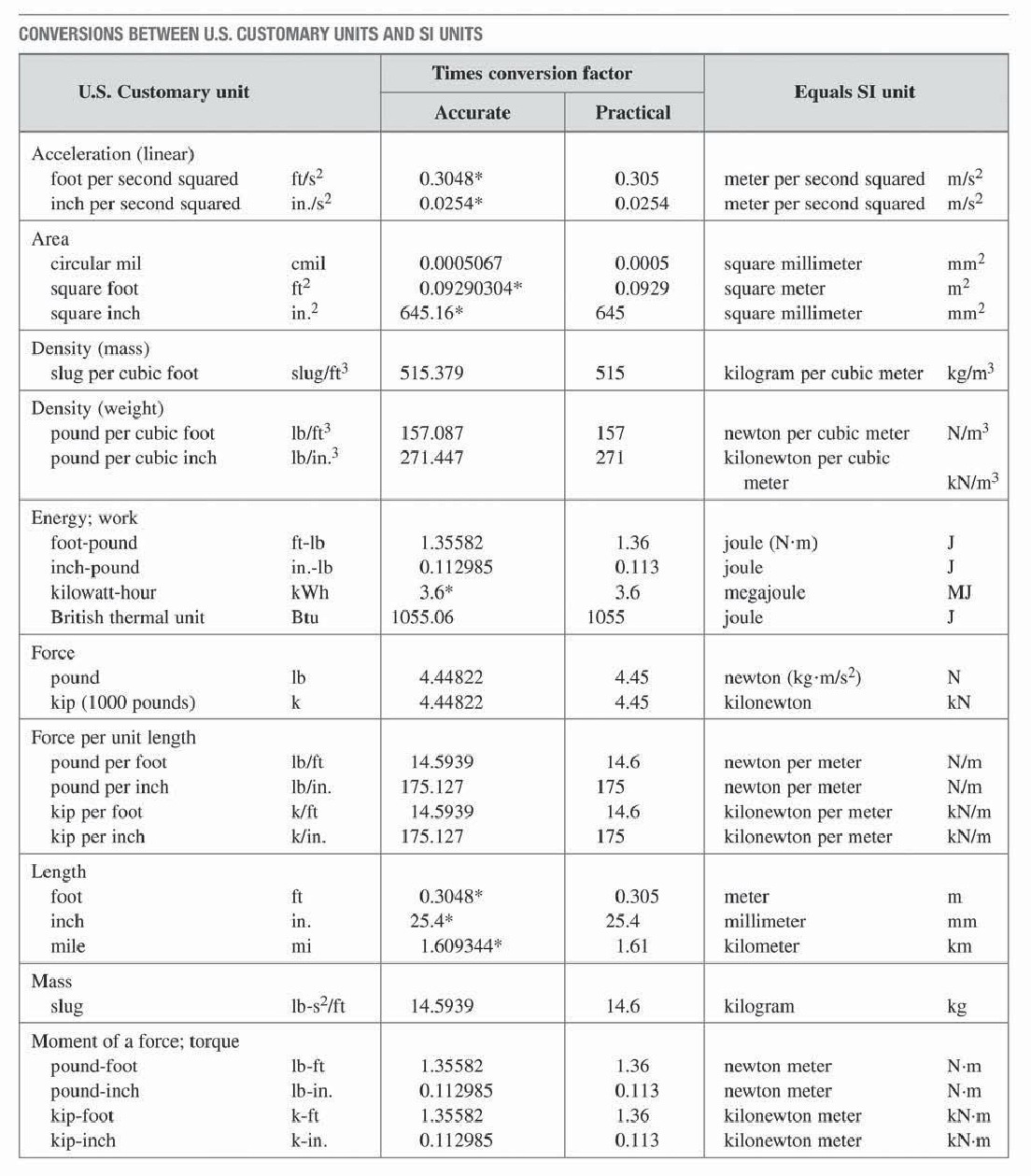
It is the resistance between two points that produces a potential difference of one volt when a current of one ampere flows between them, i.e., Ω = V/A = kg m 2/A 2 s 3. The ohm, Ω is the derived unit of electrical resistance. It is that difference in potential which generates one watt of energy per ampere of current, i.e., V = W/A = kg m 2/A s 3. The volt, V, is the derived unit of electrical potential, or potential difference. It is the quantity of electricity transported per second by a current of one ampere, i.e., C = A s. The coulomb, C, is the derived unit of electric charge. This is because of the fundamental relationship between electricity and magnetism. The only additional SI base unit involved in both electrical and magnetic quantities is the ampere, A. The entities must be specified, as atoms, molecules, ions, electrons or other particles or groups of particles.ĭerived SI units involving electricity and magnetism The mole, mol, is the amount of substance which contains as many elementary entities as there are atoms in 0.012 kilogram of the carbon isotope (Alternatively, a candela is the luminous intensity of a source that emits, in a given direction, monochromatic radiation of frequency 540 × 10 12 hertz and has a radiant intensity in the same direction of 1/683 watt per steradian.) The candela, cd, is the luminous intensity, in the perpendicular direction, of a surface 1/600,000 square meter of a full radiator at the temperature of freezing platinum under a pressure of 101,325 newtons per square meter. The kelvin, K, unit of thermodynamic temperature is the fraction 1/273.16 of the thermodynamic temperature of the triple point of water. The ampere, A, is that constant current which, if maintained in each of two infinitely long straight parallel wires of negligible crosssection, placed one meter apart in vacuum, will produce between the wires a force of 2 × 10 −7 newtons per meter length. Alternatively, the ephemeris second is defined as 1/31,556,925.974 7 of the tropical year for 1900. The second, s, is the time taken for 9,192,631,770 cycles of the radiation from the hyperfine transition in Caesium 133 when unperturbed by external fields. The Sèvres cylinder is still the standard kilogram. A platinum-iridium cylinder at Sèvres, intended to have exactly this mass, was made oversize by 28 parts per million, so for a while prior to 1964, the liter was defined at 1.000028 × 10 −3 m 3 to avoid this anomaly. The kilogram, kg, was originally defined as the mass of a liter (10 −3 m 3) of pure water at its maximum density. Alternatively a meter is the distance travelled by light in vacuum in 1/299,792,458 of a second. Today with the need for greater precision, the meter is defined as 1,650,763.73 times the wavelength, in vacuum, of the orange light emitted by This proved an inexact measure, so for a while an actual bar of platinum-iridium at Sèvres became the standard meter. 
In other words, the shortest distance from equator to pole was to be exactly 10 7 m. 
In an era of idealism it was designed to relate neatly to the size of the earth, designated as one part in forty million of the earth's circumference. The meter, m, (from the Latin metrum-measure) was introduced in France at the time of the revolution. Mole, mol, the standard of amount of substance. In the SI system there are seven base units, from which others are derived by combination, and two supplementary units that are angles.Īmper, A, the standard of electric current Ĭandela, cd, the standard of luminar intensity Système Internationale d'Unités, or SI Units, was the outcome.įor a complete detailed account of SI units and their proper usage the chapter in the Heat Exchanger Design Handbook entitled "Conventions and nomenclature for physical quantities, units numbers and mathematics" by Y. In 1948, following a resolution of the Ninth Conference of Weights and Measures (CGPM) an international committee was established to formulate a new international system of units. Over past centuries several different systems of units have been used by engineers and scientists, necessitating a large number of conversion factors and often leading to confusion. The number represents the ratio of the magnitude of the quantity (mass M) to that of the unit (kilogram, kg). When expressing the magnitude of a physical quantity, we use a number followed by a unit, e.g., M = 6 kg. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
